12 min read • Healthcare & life sciences
Changing gears to deliver CAR-T in your hospital
Enabling operational readiness for CAR-T therapy delivery in a hospital


Executive Summary
New advanced therapies, such as chimeric antigen receptor T-cell (CAR-T), are remarkable treatments for patients who have exhausted all other therapy options. These innovative therapies are delivered through non-traditional models and involve lengthy, complex, highly coordinated sets of activities within the healthcare provider’s facilities. For providers, these therapies offer a number of opportunities, from improving their stature as leading-edge institutions to offering high-revenuegenerating services. However, if not thoughtfully considered and appropriately planned, delivering these new advanced therapies presents tremendous financial and operational risks. This report sets out the strategic and readiness evaluation framework providers need to evaluate CAR-T therapy as a treatment to be offered.
2
Providers are excited about CAR-T
Given the success of CAR-T therapy, there has been a lot of interest from healthcare providers in delivering this treatment. There are three main reasons why many healthcare providers are considering the opportunity that CAR-T therapy provides: remission, return, and renown.
Remission
As therapies go, CAR-T therapy has shown to be very effective in treating hematological cancers. Complete remission rates as high as 90 percent were seen in clinical trials for patients with relapsed and refractory B-cell acute lymphoblastic leukemia, and response rates greater than 50 percent have been seen in heavily pretreated chronic lymphocytic leukemia and nonHodgkin lymphomas3 . This is a major opportunity for healthcare providers, as these new therapies support what providers are very good at – delivery of improved health outcomes for patients.More than 80 percent of children diagnosed with Acute Lymphoblastic Leukemia (ALL) can achieve long-term event-free survival (EFS) with existing treatments. However, for patients whose cancers return after chemotherapy or a stem cell transplant, there have been few or no treatment options. Novartis was the first company to generate impressive clinical data and subsequently launch its CAR-T therapy product, Kymriah, a one-time treatment for B-cell ALL. Clinical trial data demonstrated an 83 percent remission rate after three months in patients who had not responded to standard treatments.
Return
Not all new therapies can support the delivery of improved health outcomes at an acceptable cost. According to published literature4 , there has been a trend of negligible incremental effectiveness, despite the rising cost per life year gained from recent cancer therapies. Research published and discussed at the 2018 annual meeting of the American Society of Hematology indicates that CAR-T therapy goes against the trend seen with recent hematology therapies, as it provides improvement in health outcomes (measured in quality-adjusted life years) to a degree higher than that from treatments for nonhematologic cancers and non-CAR-T treatments for hematologic cancers.
Renown
There are extended benefits for hospitals that develop the capability to provide CAR-T therapy to patients. They have the potential to attract clinicians who are keen to develop their competence with CAR-T therapy and would otherwise not have considered these hospitals as places to further their careers. In addition, existing talent in the hospital can develop competency for delivery of a cutting-edge medical intervention to patients. Furthermore, the opportunity to be a national or global research center of choice, and to publish research papers from firsthand clinical experience in an emerging treatment area, will make the hospital a desirable career destination for hematologists and oncologists.
3
Due diligence should precede investment
The investment case has to be carefully considered
The decision to invest resources to support the delivery of ACT-based therapies requires careful evaluation by healthcare providers. Basic considerations such as whether there are sufficient numbers of patients to justify the investment have to be made. Not only are there significant upfront infrastructure costs, but an entirely new model of work is required to operationalize the delivery of treatments like CAR-T therapy. Examples include process and eligibility criteria to ensure that patients with the most need are selected, laboratories and facilities to collect and process the required cells, quality management systems, inventory systems, and care infrastructure to manage patients during cell processing and following infusion.
Establishing CAR-T and ACT-based services in a hospital generally begins with development of an initial high-level business case. As with any new therapy, business model parameters must be rigorously considered since specific data is still unavailable (or emerging). The business case will have to rely on many assumptions due to lack of information on the operational complexities of the service delivery. While the high-level business case is vital in the decision-making process, taking it on its own without further analysis of the detailed set-up requirements of a CAR-T service will be reckless. It is therefore imperative to understand in detail the operational requirements, gaps in a hospital’s infrastructure, and what is needed to bridge the gap before finalizing the business case or building an implementation roadmap for CAR-T therapy.
Figure 1: CAR-T treatment pathway
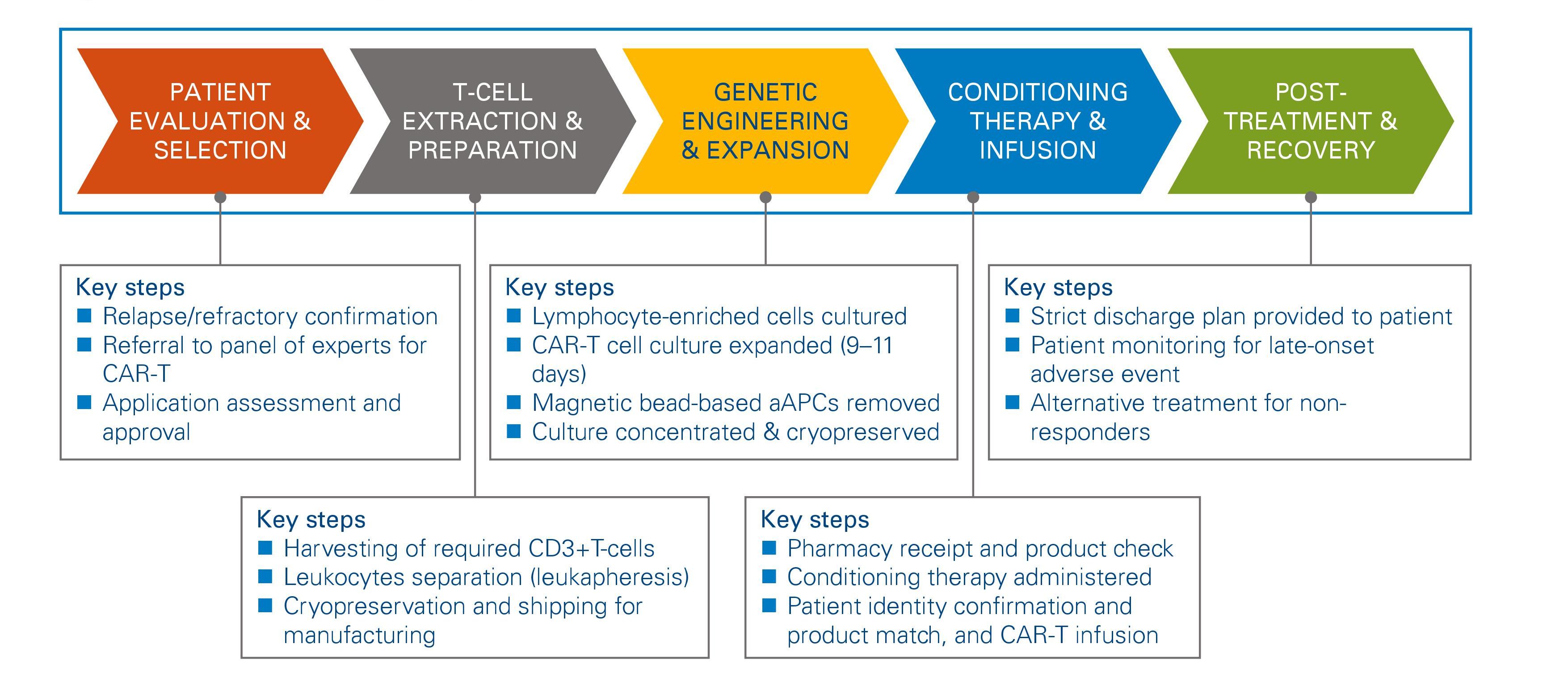
Stay on the right path
One of the best ways to assess the operational readiness of a provider to introduce CAR-T therapy is to use the treatment pathway as a framework to understand the infrastructure needed to deliver the service. The treatment pathway is ideal because it is an integrated, end-to-end view that considers the needs of the patients, manufacturer, provider, and payer. It provides a comprehensive view – from the moment the patient is assessed for eligibility to the post-treatment phase. Applied robustly, the treatment pathway will highlight the major requirements and gaps within a provider’s existing set-up, as well as provide vital information to strengthen the initial business case.
4
Follow the treatment pathway
From a high-level view, the CAR-T treatment pathway consists of five main steps:
Patient evaluation and selection
The first step spans the patient journey from the relapse/ refractory phase of previous therapies through to selection and approval for CAR-T therapy. This includes the battery of tests required to confirm relapse and eligibility for CAR-T therapy, the application for approval sent to the governing panel (a local and/or national panel of cancer experts), and approval or authorization to proceed with the therapy. It also involves the provision of alternative treatments for patients who are not eligible or whose applications are unsuccessful.
T-cell extraction and preparation
The next step in the framework involves the necessary activities and procedures required to extract the required T-cells from a patient’s blood – a process called leukapheresis. Ideally, leukapheresis is optimized to generate sufficient materials in one session. It involves collecting a sample of the patient’s blood and extracting the lymphocytes, before returning the remaining blood to the patient’s bloodstream. This step also involves labeling and cryopreservation of the sample, processing the required paperwork for transportation of the sample to the manufacturing center, and linking the hospital’s system with the logistics system of the manufacturer in order to ensure electronic tracking of the order, processing, and delivery.
Genetic engineering and expansion
At this step, the harvested T-cells are genetically modified to express the chimeric antigen receptor, and subsequently expanded in vitro until enough CAR-T cells are manufactured for effective treatment. This process takes a few weeks and requires coordinated communication with the medical center treatment team, in order to ensure that the patient receives interim treatment or initiate any therapy washouts. Finally, the culture is then cryopreserved and returned to the treatment center. Currently, for approved treatments, the patient sample has to be sent off to contract manufacturers, who carry out genetic engineering and expansion. There are emerging approaches, currently in clinical trials, which may allow hospitals to do genetic engineering and expansion.
Lymphodepletion conditioning therapy
At this stage, the CAR-T cells are infused into the patient, who is then carefully monitored within the treatment facility. Prior to infusion, it is common for the patient to be given a dose of chemotherapy and other lymphodepletion conditioning therapy to maximize the therapeutic effect of the treatment. Strict quality control checks are also implemented to ensure that the right product is given to the right patient. Finally, reinfusion might also be required, depending on the success of the first CAR-T cell infusion.
Post-treatment and recovery
The final step of the CAR-T treatment journey involves assessment of the therapeutic and adverse effects of the therapy before the patient is discharged. The assessment includes monitoring the patient for toxicity effects of the therapy and, as appropriate, management of any side effects. This step also involves significant education of the patient and caregiver, particularly on possible late-onset adverse events. The patient often requires remote monitoring post-discharge and is expected to reside within a two-hour driving radius of the treatment center for a number of weeks. Finally, the treating hospital is required to provide post-therapy reports to the appropriate health authority.
5
Do a thorough gap assessment
Providers planning to add CAR-T therapy to their existing services will need to assess the gaps in the treatment pathways in their hospitals, using a gap assessment framework.
Governance and guidelines
This involves obtaining the necessary accreditations and certifications from the governing bodies and manufacturers to provide this service. It also involves establishing the governance required to assess and approve individual CAR-T therapy request applications. It is be very important for providers to decide whether they want to be involved in product manufacturing as this will influence what is required from a governance perspective. Some providers might need to form new panels of experts or augment the scope of existing suitable panels to provide the required governance for their hospitals. In some countries, governance may extend beyond the local (hospital) level to a regional or national review panel.
Figure 2: CAR-T treatment pathway-based gap assessment framework
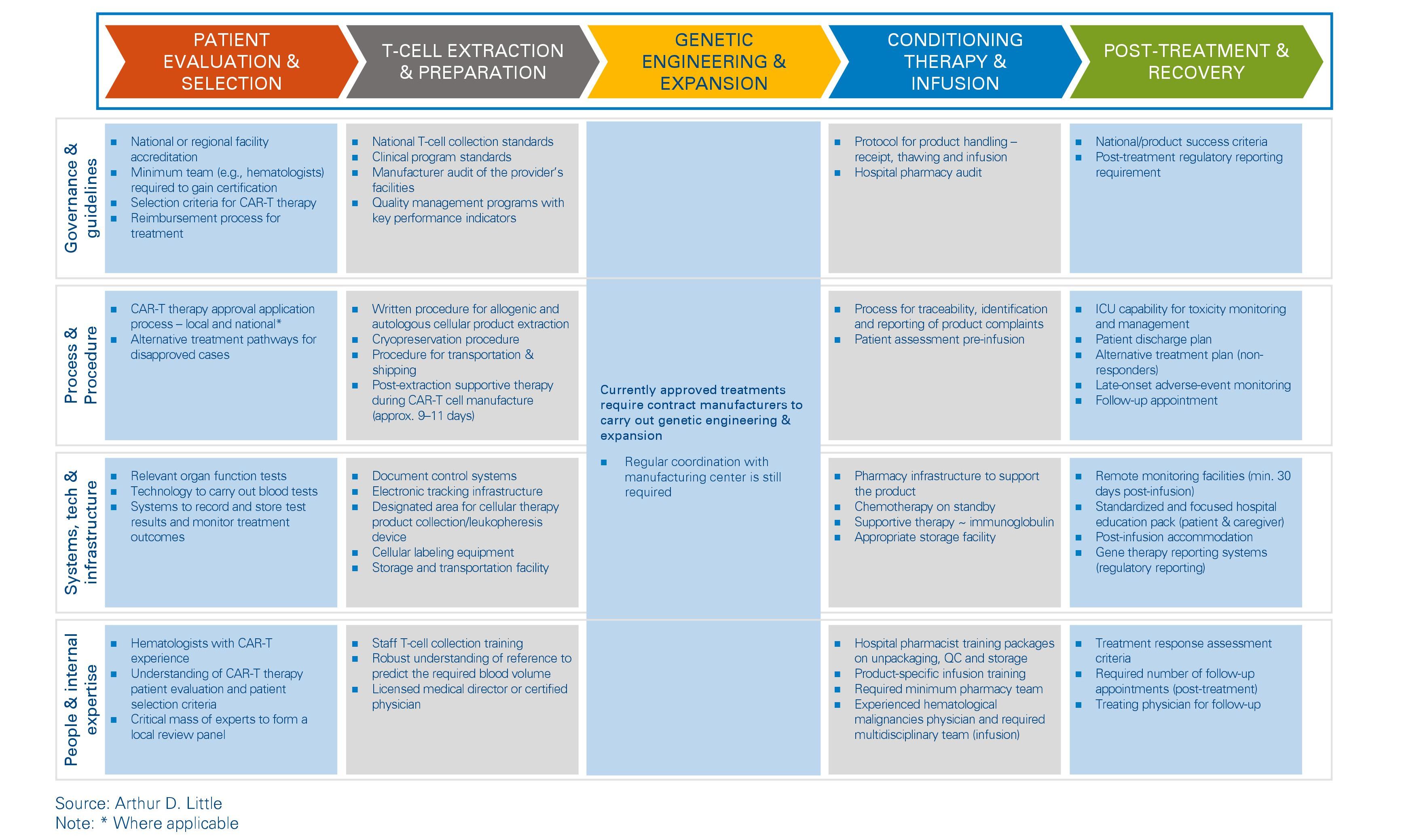
CAR-T is an unconventional therapy, and as such, the guidelines for therapy (from T-cell extraction and labeling to genetic modification and reinfusion into the patient) may not exist. These guidelines, which could be local, national and/or product specific, will need to be established.
Processes and procedures
Assessing the internal processes required to deliver CAR-T therapy could reveal gaps such as lack of a therapy application process or the absence of a process for labeling and storage of the culture. Upholding the high-quality and process standards for this therapy requires either creation of new processes or strengthening of existing processes. These are also subject to GxP audits. Medical procedures specific to CAR-T therapy may also be an area with gaps. An example could be absence of processes for the supportive therapy that must be administered during the weeks of CAR-T cell manufacture.
Systems and infrastructure
Additional control systems could be required to supplement the manual effort involved in the therapy delivery process and minimize human errors – which could be very costly with this therapy. Examples include robust data and document control systems, overarching quality management systems and cellular labeling equipment, and pharmacy controls and checks.
This category also includes facilities (e.g., appropriate designated areas for collection and storage of cellular therapy products) needed to smoothly run the operation and ensure patient safety and effective therapy. A gap assessment here involves review of the infrastructure required to link the hospital’s system with the logistics systems of the manufacturer so ordering, processing, and delivery can be tracked and completed electronically.
People and internal expertise
As with introduction of any new advanced therapy, additional expertise will be required to enable successful implementation. This includes upskilling of existing staff members (e.g., hospital pharmacist training packages on unpackaging, QC and storage of CAR-T culture) and the associated changes to their job descriptions. In most cases, additional external personnel will be needed to provide expertise that is not available within the hospital (e.g., a product-certified physician) to cover the additional work demand. Assessing the necessary end-to-end expertise and comparing it to the current internal proficiency will highlight strengths and significant shortfalls.
6
Change gears with CAR-T therapy
CAR-T therapy is an exciting development to add to a healthcare provider’s arsenal and give hope to patients who would otherwise be left with no real alternatives. However, hospitals will have to move quickly if there is real interest in developing the capability to deliver it. This is because the number of patients that will qualify for CAR-T therapy is so low that it will not make investment sense to develop the capability if another provider in the geographical region is already providing the service. For the foreseeable future, while the technology will still be expensive, CAR-T therapy will only be delivered by hospitals that are motivated to quickly establish themselves as the providers of the service in their regions. That journey begins by developing a high-level business case and using the treatment pathway framework to assess operational readiness and uncover the delivery gaps. This can assist in developing a more detailed business case. Once the investment picture is clear and the returns are considered compelling, it will be time to change gears and begin the journey to establish your hospital in a higher league of healthcare providers.
DOWNLOAD THE FULL REPORT
12 min read • Healthcare & life sciences
Changing gears to deliver CAR-T in your hospital
Enabling operational readiness for CAR-T therapy delivery in a hospital

DATE

Executive Summary
New advanced therapies, such as chimeric antigen receptor T-cell (CAR-T), are remarkable treatments for patients who have exhausted all other therapy options. These innovative therapies are delivered through non-traditional models and involve lengthy, complex, highly coordinated sets of activities within the healthcare provider’s facilities. For providers, these therapies offer a number of opportunities, from improving their stature as leading-edge institutions to offering high-revenuegenerating services. However, if not thoughtfully considered and appropriately planned, delivering these new advanced therapies presents tremendous financial and operational risks. This report sets out the strategic and readiness evaluation framework providers need to evaluate CAR-T therapy as a treatment to be offered.
2
Providers are excited about CAR-T
Given the success of CAR-T therapy, there has been a lot of interest from healthcare providers in delivering this treatment. There are three main reasons why many healthcare providers are considering the opportunity that CAR-T therapy provides: remission, return, and renown.
Remission
As therapies go, CAR-T therapy has shown to be very effective in treating hematological cancers. Complete remission rates as high as 90 percent were seen in clinical trials for patients with relapsed and refractory B-cell acute lymphoblastic leukemia, and response rates greater than 50 percent have been seen in heavily pretreated chronic lymphocytic leukemia and nonHodgkin lymphomas3 . This is a major opportunity for healthcare providers, as these new therapies support what providers are very good at – delivery of improved health outcomes for patients.More than 80 percent of children diagnosed with Acute Lymphoblastic Leukemia (ALL) can achieve long-term event-free survival (EFS) with existing treatments. However, for patients whose cancers return after chemotherapy or a stem cell transplant, there have been few or no treatment options. Novartis was the first company to generate impressive clinical data and subsequently launch its CAR-T therapy product, Kymriah, a one-time treatment for B-cell ALL. Clinical trial data demonstrated an 83 percent remission rate after three months in patients who had not responded to standard treatments.
Return
Not all new therapies can support the delivery of improved health outcomes at an acceptable cost. According to published literature4 , there has been a trend of negligible incremental effectiveness, despite the rising cost per life year gained from recent cancer therapies. Research published and discussed at the 2018 annual meeting of the American Society of Hematology indicates that CAR-T therapy goes against the trend seen with recent hematology therapies, as it provides improvement in health outcomes (measured in quality-adjusted life years) to a degree higher than that from treatments for nonhematologic cancers and non-CAR-T treatments for hematologic cancers.
Renown
There are extended benefits for hospitals that develop the capability to provide CAR-T therapy to patients. They have the potential to attract clinicians who are keen to develop their competence with CAR-T therapy and would otherwise not have considered these hospitals as places to further their careers. In addition, existing talent in the hospital can develop competency for delivery of a cutting-edge medical intervention to patients. Furthermore, the opportunity to be a national or global research center of choice, and to publish research papers from firsthand clinical experience in an emerging treatment area, will make the hospital a desirable career destination for hematologists and oncologists.
3
Due diligence should precede investment
The investment case has to be carefully considered
The decision to invest resources to support the delivery of ACT-based therapies requires careful evaluation by healthcare providers. Basic considerations such as whether there are sufficient numbers of patients to justify the investment have to be made. Not only are there significant upfront infrastructure costs, but an entirely new model of work is required to operationalize the delivery of treatments like CAR-T therapy. Examples include process and eligibility criteria to ensure that patients with the most need are selected, laboratories and facilities to collect and process the required cells, quality management systems, inventory systems, and care infrastructure to manage patients during cell processing and following infusion.
Establishing CAR-T and ACT-based services in a hospital generally begins with development of an initial high-level business case. As with any new therapy, business model parameters must be rigorously considered since specific data is still unavailable (or emerging). The business case will have to rely on many assumptions due to lack of information on the operational complexities of the service delivery. While the high-level business case is vital in the decision-making process, taking it on its own without further analysis of the detailed set-up requirements of a CAR-T service will be reckless. It is therefore imperative to understand in detail the operational requirements, gaps in a hospital’s infrastructure, and what is needed to bridge the gap before finalizing the business case or building an implementation roadmap for CAR-T therapy.
Figure 1: CAR-T treatment pathway

Stay on the right path
One of the best ways to assess the operational readiness of a provider to introduce CAR-T therapy is to use the treatment pathway as a framework to understand the infrastructure needed to deliver the service. The treatment pathway is ideal because it is an integrated, end-to-end view that considers the needs of the patients, manufacturer, provider, and payer. It provides a comprehensive view – from the moment the patient is assessed for eligibility to the post-treatment phase. Applied robustly, the treatment pathway will highlight the major requirements and gaps within a provider’s existing set-up, as well as provide vital information to strengthen the initial business case.
4
Follow the treatment pathway
From a high-level view, the CAR-T treatment pathway consists of five main steps:
Patient evaluation and selection
The first step spans the patient journey from the relapse/ refractory phase of previous therapies through to selection and approval for CAR-T therapy. This includes the battery of tests required to confirm relapse and eligibility for CAR-T therapy, the application for approval sent to the governing panel (a local and/or national panel of cancer experts), and approval or authorization to proceed with the therapy. It also involves the provision of alternative treatments for patients who are not eligible or whose applications are unsuccessful.
T-cell extraction and preparation
The next step in the framework involves the necessary activities and procedures required to extract the required T-cells from a patient’s blood – a process called leukapheresis. Ideally, leukapheresis is optimized to generate sufficient materials in one session. It involves collecting a sample of the patient’s blood and extracting the lymphocytes, before returning the remaining blood to the patient’s bloodstream. This step also involves labeling and cryopreservation of the sample, processing the required paperwork for transportation of the sample to the manufacturing center, and linking the hospital’s system with the logistics system of the manufacturer in order to ensure electronic tracking of the order, processing, and delivery.
Genetic engineering and expansion
At this step, the harvested T-cells are genetically modified to express the chimeric antigen receptor, and subsequently expanded in vitro until enough CAR-T cells are manufactured for effective treatment. This process takes a few weeks and requires coordinated communication with the medical center treatment team, in order to ensure that the patient receives interim treatment or initiate any therapy washouts. Finally, the culture is then cryopreserved and returned to the treatment center. Currently, for approved treatments, the patient sample has to be sent off to contract manufacturers, who carry out genetic engineering and expansion. There are emerging approaches, currently in clinical trials, which may allow hospitals to do genetic engineering and expansion.
Lymphodepletion conditioning therapy
At this stage, the CAR-T cells are infused into the patient, who is then carefully monitored within the treatment facility. Prior to infusion, it is common for the patient to be given a dose of chemotherapy and other lymphodepletion conditioning therapy to maximize the therapeutic effect of the treatment. Strict quality control checks are also implemented to ensure that the right product is given to the right patient. Finally, reinfusion might also be required, depending on the success of the first CAR-T cell infusion.
Post-treatment and recovery
The final step of the CAR-T treatment journey involves assessment of the therapeutic and adverse effects of the therapy before the patient is discharged. The assessment includes monitoring the patient for toxicity effects of the therapy and, as appropriate, management of any side effects. This step also involves significant education of the patient and caregiver, particularly on possible late-onset adverse events. The patient often requires remote monitoring post-discharge and is expected to reside within a two-hour driving radius of the treatment center for a number of weeks. Finally, the treating hospital is required to provide post-therapy reports to the appropriate health authority.
5
Do a thorough gap assessment
Providers planning to add CAR-T therapy to their existing services will need to assess the gaps in the treatment pathways in their hospitals, using a gap assessment framework.
Governance and guidelines
This involves obtaining the necessary accreditations and certifications from the governing bodies and manufacturers to provide this service. It also involves establishing the governance required to assess and approve individual CAR-T therapy request applications. It is be very important for providers to decide whether they want to be involved in product manufacturing as this will influence what is required from a governance perspective. Some providers might need to form new panels of experts or augment the scope of existing suitable panels to provide the required governance for their hospitals. In some countries, governance may extend beyond the local (hospital) level to a regional or national review panel.
Figure 2: CAR-T treatment pathway-based gap assessment framework

CAR-T is an unconventional therapy, and as such, the guidelines for therapy (from T-cell extraction and labeling to genetic modification and reinfusion into the patient) may not exist. These guidelines, which could be local, national and/or product specific, will need to be established.
Processes and procedures
Assessing the internal processes required to deliver CAR-T therapy could reveal gaps such as lack of a therapy application process or the absence of a process for labeling and storage of the culture. Upholding the high-quality and process standards for this therapy requires either creation of new processes or strengthening of existing processes. These are also subject to GxP audits. Medical procedures specific to CAR-T therapy may also be an area with gaps. An example could be absence of processes for the supportive therapy that must be administered during the weeks of CAR-T cell manufacture.
Systems and infrastructure
Additional control systems could be required to supplement the manual effort involved in the therapy delivery process and minimize human errors – which could be very costly with this therapy. Examples include robust data and document control systems, overarching quality management systems and cellular labeling equipment, and pharmacy controls and checks.
This category also includes facilities (e.g., appropriate designated areas for collection and storage of cellular therapy products) needed to smoothly run the operation and ensure patient safety and effective therapy. A gap assessment here involves review of the infrastructure required to link the hospital’s system with the logistics systems of the manufacturer so ordering, processing, and delivery can be tracked and completed electronically.
People and internal expertise
As with introduction of any new advanced therapy, additional expertise will be required to enable successful implementation. This includes upskilling of existing staff members (e.g., hospital pharmacist training packages on unpackaging, QC and storage of CAR-T culture) and the associated changes to their job descriptions. In most cases, additional external personnel will be needed to provide expertise that is not available within the hospital (e.g., a product-certified physician) to cover the additional work demand. Assessing the necessary end-to-end expertise and comparing it to the current internal proficiency will highlight strengths and significant shortfalls.
6
Change gears with CAR-T therapy
CAR-T therapy is an exciting development to add to a healthcare provider’s arsenal and give hope to patients who would otherwise be left with no real alternatives. However, hospitals will have to move quickly if there is real interest in developing the capability to deliver it. This is because the number of patients that will qualify for CAR-T therapy is so low that it will not make investment sense to develop the capability if another provider in the geographical region is already providing the service. For the foreseeable future, while the technology will still be expensive, CAR-T therapy will only be delivered by hospitals that are motivated to quickly establish themselves as the providers of the service in their regions. That journey begins by developing a high-level business case and using the treatment pathway framework to assess operational readiness and uncover the delivery gaps. This can assist in developing a more detailed business case. Once the investment picture is clear and the returns are considered compelling, it will be time to change gears and begin the journey to establish your hospital in a higher league of healthcare providers.
DOWNLOAD THE FULL REPORT

